
(a) If a soft-drink bottle whose volume is 1. The answer is rounded to the nearest tenth, since the mass of the water was known to the nearest tenth (the last operation involves addition, so we pay attention to decimal places when rounding rather than significant figures). This widget calculates the density of water at various temperatures and pressures. Water has a density of 0.997 g>cm3 at 25 C ice has a density of 0.917 g>cm3 at -10 C. Direct measurement for density of seawater is done by maintaining the collected water samples in controlled environmental conditions.\) Likewise, check the water density of samples having same salinity and different temperatures. All you need is enter the temperature (both in Celsius and Fahrenheit) and salinity in PPM (parts per million) or mg/L in the suggested fields.Īfter finding the results, you can correlate the density of water at room temperature for different salinity levels. If you are interested in calculating the density of seawater at different temperatures, there are online tools that are developed for the same purpose. Of course, this is true for other substances as well. When water is cooled at room temperature, it becomes denser over time. Water - Density, Specific Weight and Thermal Expansion Coefficients. Temperature can also change water density, but the correlation is still unclear and inconsistent. Thus, water with lower salt content will float over water with higher salt concentration. The density of water is approximately 1 gram per cubic centimeter (1 g/cm3) or 1 gram per milliliter (1g/mL). In short, density of water is directly proportional to salinity. As an example of how water density changes with temperature, at 1 atm pressure water has an estimated density of 983.854 kg/m 3 at -30 ☌, 999. In general, the density of a matter is higher in solid form, as compared to the density of the same matter in liquid form.Īs with salinity, higher the level of salt concentration, the more will be the density of water, even at room temperature. However, as we are discussing the density of water measured in ground level for collected water samples, the pressure attribute is not considered in determining the density. 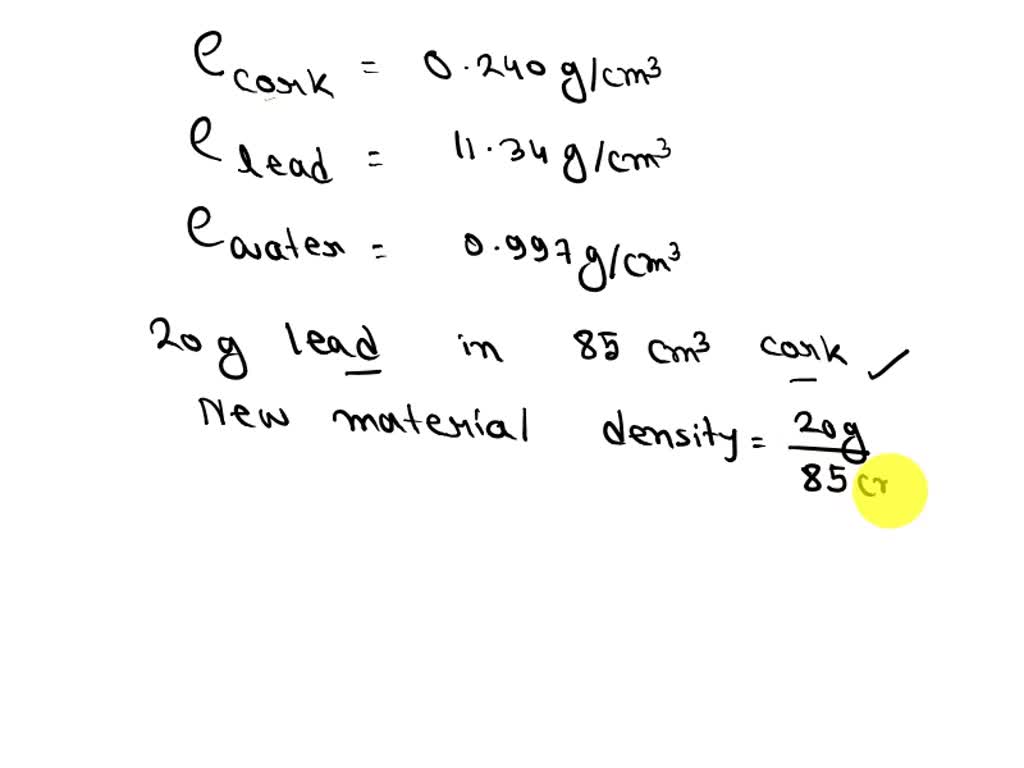
Water displacement works because the amount of water displaced by an object submerged in water equals the. Find mass using a balance scale, and use water displacement to find the volume of irregular objects. 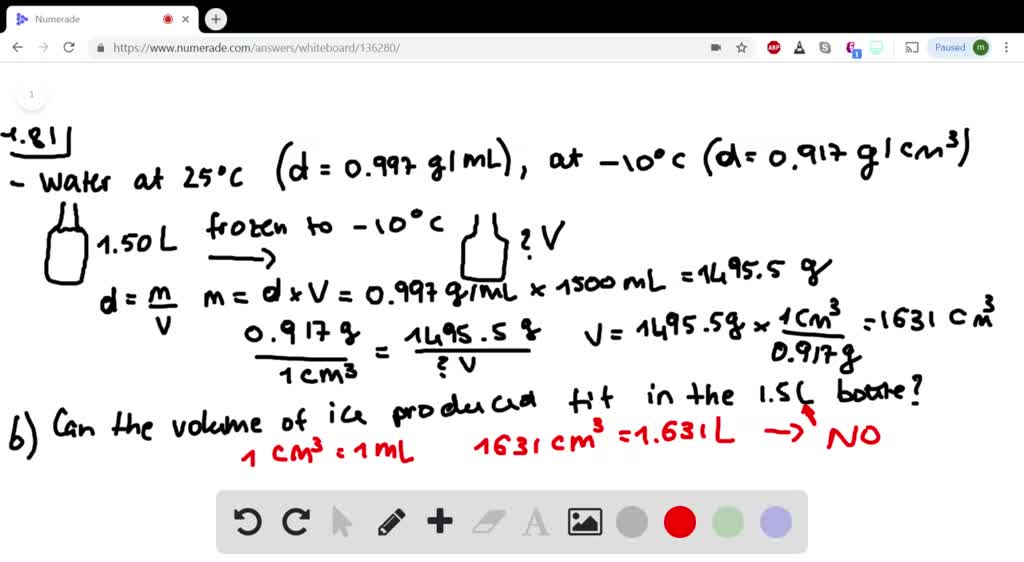
Of course, pressure does play a role in the subject of water denseness. Density (g/cm3) at 20C and 1 atm unless noted water 0.99820 water (0 C) 0.99984 water (4 C) 0.99997 water (100 C) 0.95836 gasoline 0.66-0.69 ethyl alcohol 0.791 turpentine 0.85 olive oil 0.9 castor oil 0.969 sea water: 1.03 milk 1.028-1.035 glycerin 1.260 mercury 13. TL DR (Too Long Didnt Read) Calculating density uses the formula D m ÷ v, where D means density, m means mass and v means volume. While discussing this topic in detail, water density is found to vary with respect to the level of salinity (or other impurities) and fluctuation of temperature. However, it is to be noted that grams per milliliter (g/mL) and gram per cubic centimeter (g/cm 3) are not SI units. The logic is to divide the value of kg/m 3 by 1000 to get pure water density in g/ml. In other words, at the same temperature, the density of water in g/ml (or g/cm 3) is 0.99777. At room temperature (i.e., 22° C), the density of water in kg/m 3 is 997.77. With respect to the definition, the density formula is represented as Density (ρ) = Mass (m)/Volume (V).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
